AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Niels bohr's model of the hydrogen atom3/16/2024  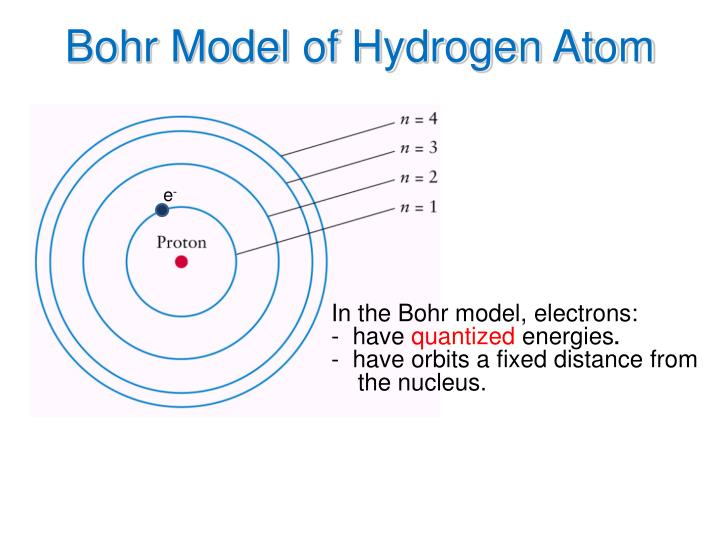
In this picture, each orbit is associated with a particular energy, and the electron can change orbit by emitting or absorbing energy in discrete chunks (called quanta). Rutherford’s model didn’t account for the stability of atoms, so Bohr turned to the burgeoning field of quantum physics, which deals with the microscopic scale, for answers.īohr suggested that instead of buzzing randomly around the nucleus, electrons inhabit orbits situated at a fixed distance away from the nucleus. According to the rules of classical physics, the electrons would eventually spiral down into the nucleus, causing the atom to collapse. The dense nucleus is surrounded by a swarm of tiny, negatively charged electrons.īohr, who worked for a key period in 1912 in Rutherford’s laboratory in Manchester in the UK, was worried about a few inconsistencies in this model. The vast majority of its mass is located in a positively charged central nucleus, which is 10,000 times smaller than the atom itself. 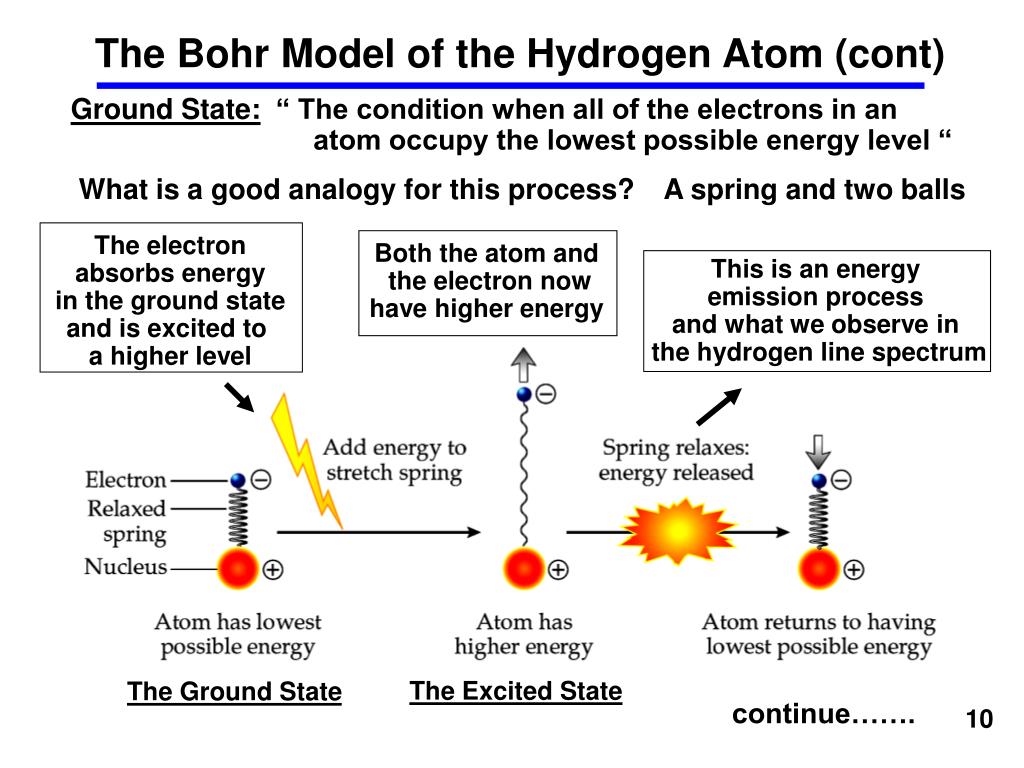
Rutherford had made the startling discovery that most of the atom is empty space. In July of 1913, Danish physicist Niels Bohr published the first of a series of three papers introducing this model of the atom, which became known simply as the Bohr atom.īohr, one of the pioneers of quantum theory, had taken the atomic model presented a few years earlier by physicist Ernest Rutherford and given it a quantum twist. 
The most instantly recognizable image of an atom resembles a miniature solar system with the concentric electron paths forming the planetary orbits and the nucleus at the centre like the sun. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
